PAYMENT
We accept payment via PayPal. However if you don’t have Paypal account you can directly pay with your credit card to our PayPal account
SHIPPING WORLD-WIDE
Mygifts360 Shop do shipping world-wide via trusted and reliable logistics company
QUALITY ASSURANCE
We only sell 100% genuine goods and products with high quality.
NEED HELP?
We are here to help you 24/7. Connect with us on Facebook or email us at support@mygifts360.com
-
SAVORY OLIVE GIFT BASKET
$139.99
FEATURED PRODUCTS
-
Bear Case Dollhouse Wood Plastic Dolls Houses
Original price was: $48.92.$26.96Current price is: $26.96. -
SNACK GIFT BASKET – PREMIUM
$94.99 -
Spring Infant Baby Cartoon Clothing Sets
$10.44 – $17.28 -
Trendy Ins Clavicle Chain Valentines Day Gift Party Jewelry
Original price was: $23.94.$12.95Current price is: $12.95.
Best Selling Products
-
Waterproof Wireless Bluetooth Headphones
$16.70 – $27.20 -
Magnetic USB Fast Charging Micro / Type C Cable
$8.99 – $9.95 -
USB Cable for iPhone
$8.99 – $9.95
Recently Products
-
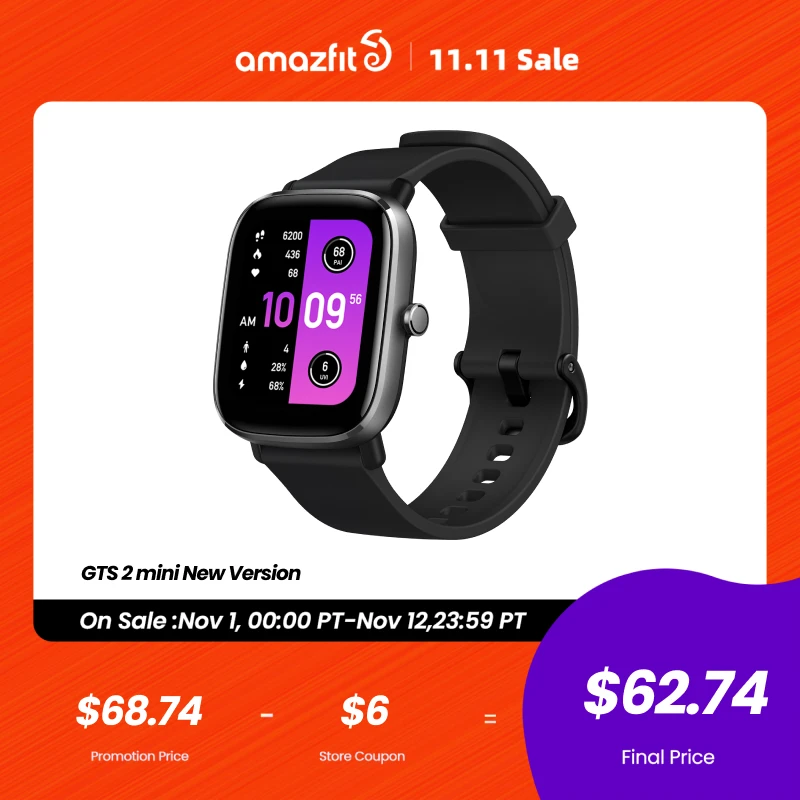
[New Version] Amazfit GTS 2 mini Smartwatch 68+Sports Modes Sleep Monitoring Smart Watch Zepp App For Android For iOS
Original price was: $156.24.$106.25Current price is: $106.25.
Products Attributes
-
Metal Balloon Arch Support Kit Outdoor Wedding Iron Circle
$36.96 – $136.36 -
Transparent Fruit and Vegetable Food Storage Basket
$14.95 – $27.12